Life Factory
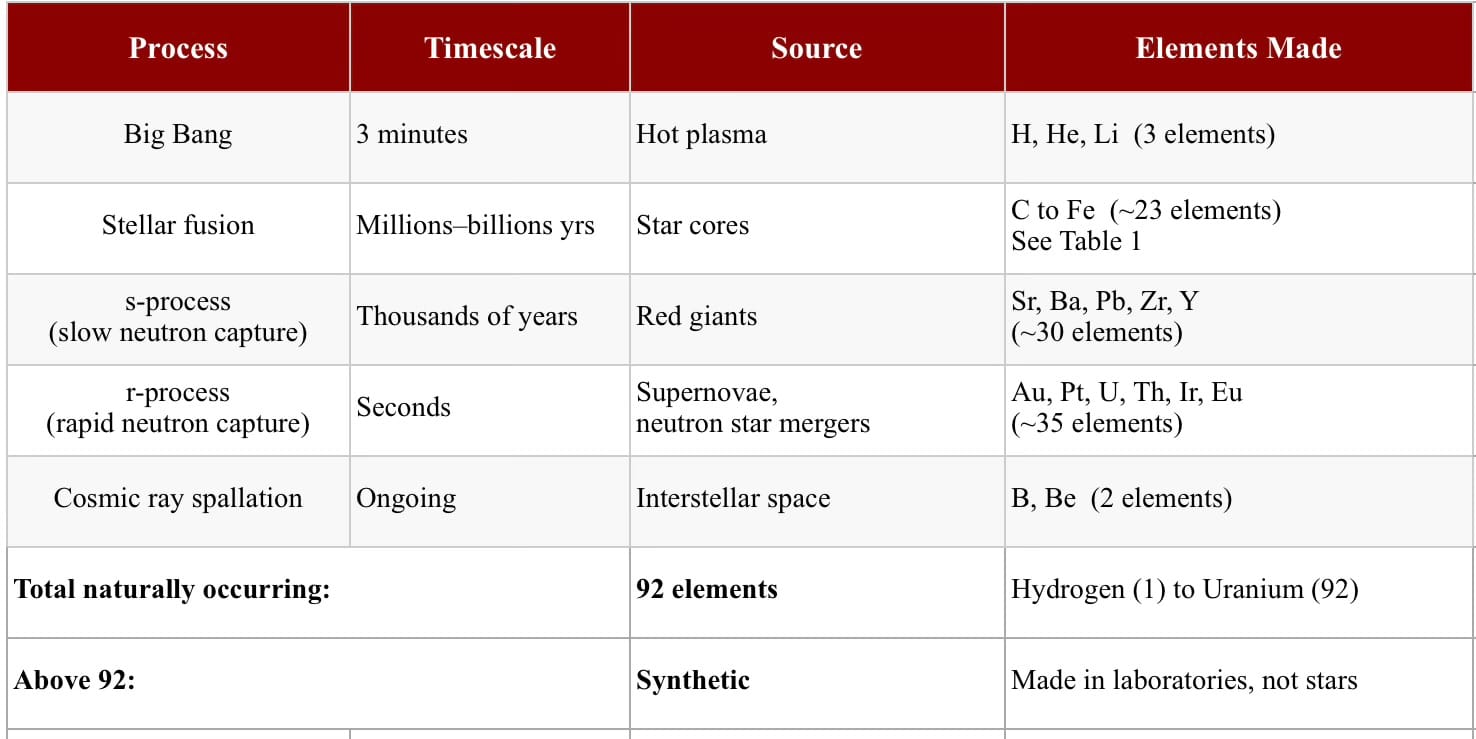
Nucleo synthesis - the material of life
The making of the nucleus
BIG BANG
t=0
Universe is a super, super hot plasma.
- Protons and neutrons form.
- Too hot for atoms — nuclei only.
∙ ~74.99% Hydrogen (1 proton)…
∙ ~24.99% Helium-4 (2 protons, 2 neutrons)…
∙ Trace Lithium-7…
∙ Trace Beryllium-7 (decays to Lithium)…
The universe cools too fast for heavier elements to form
0.007 strong nuclear coupling constant…
- it governs the binding of all nuclei…
H, He, Li
t+3 minutes
……………dark ages………400,000 years……………………
- Universe cools enough for electrons to bind to nuclei — first atoms…
- No stars yet…
- Just gas…
- And 3 elements- H, He, Li…
- Hydrogen and helium clouds drifting in darkness…
…………first stars………100 - 200 million years…………
- pi, the gear ratio of all circular motion; all motion…
- Gravity pulls hydrogen into rotating clouds, orbiting; equations based on pi…
- First Stars ignite…
- Massive - 100 times our sun…
- Compression…
- hot, bright, burns the candle at both ends…
- don’t last long…
1st Generation stars
- Stellar fusion begins...
- the element factory… 92 in total
Each element requires higher temperatures…
- produces heavier elements:

Each step up the element weight ladder releases energy, causing the star to heat up further for the next element, until
- Iron
- because fusing iron
- consumes energy instead of
- releasing it…
- The star can no longer support itself…
And goes…
BANG
- Explosive nucleosynthesis…
Supernova & neutron star mergers…
∙ r-process: rapid neutron capture
- In seconds…
- nuclei are bombarded with neutrons so fast they can’t decay between captures.
- Builds the heaviest elements:
- gold, platinum, uranium...
Red giant stars
- Before the bang, in their red giant twilight…
∙ s-process: slow neutron capture
- over thousands of years…
- Neutrons absorbed one at a time with gaps between.
- Makes some intermediate heavy elements:
- barium, lead, strontium…
2nd Generation Stars and Planets
New clouds form from the enriched debris. Enriched with the debris of first generation supernovae…
- now containing carbon, oxygen, iron, and heavier elements...
- These stars, with gravity, build planets...
- Rocky planets...
- Chemistry becomes possible...
- Molecules form...
The ingredients for life are in the mix, but not yet cooking…
3rd Generation Stars and Planets
Our sun is a third-generation star (even 4th or 5th see Appendices)…
- it contains elements that could only have been made in previous supernovae…
- the Earth condensed from the same debris cloud...
- Every element heavier than lithium in our bodies was made inside a star that died before our sun was born…
The supply chain of life…

The death of stars is for the life of us…
- they live on… through us..
Appendix 1
Our sun won’t go bang. It’s too small for a supernova…
- minimum for BANG is about 8+ solar masses…
- Our sun is a lightweight…
- one solar mass…
What will happen in about
5 billion years:
∙ Hydrogen fuel in the core runs out…
∙ Core contracts, heats up…
∙ Outer layers expand into red giant phase…
∙ Sun swells to engulf Mercury, Venus, probably Earth…
∙ Helium fusion begins in the core makes…
∙ carbon and oxygen…
∙ Outer layers puff off as a gorgeous planetary nebula…
∙ Core remains as a white dwarf…
- a dense ember of carbon and oxygen,
- cooling for eternity
- No bang.
- A sigh…
Just a slow, beautiful exhalation…of C and O… nearly co2…
Appendix 2
Astronomy actually counts star generation backwards; probably to avoid begging the question…
- “…and then what happens to ‘0’ population stars?”…
The terminology is…
Population III = first generation stars
- hydrogen and helium only, no metals…
- Never directly observed…
- Probably all massive…
- all dead…
- all exploded long ago…
- all dead…
Population II = second generation
- low metallicity, old…
- Found in galactic halos and globular clusters...
- 10-13 billion years old…
Population I = third generation +
- high metallicity, young…
- Our sun is Population I…
- Contains heavy elements from (many) previous generations’ supernovae…
- Our sun is Population I…
Our sun is considered a
- third, fourth, or possibly fifth generation star…
- based on its high heavy element content…which
- requires multiple cycles of stellar birth and…
- death to accumulate…
- requires multiple cycles of stellar birth and…
- based on its high heavy element content…which
The generation count is broad brush because gas clouds don’t always know which gen they are and get confused…as
- each new star forms from a blend of debris in the star nursery, the nebulae, from many predecessors, not a single parent star.
It’s a mulch, a pig’s breakfast, a paddy’s parliament…
Appendix 3
Lithium is element 3, one of the
- ‘ancient’ trinity, with H and He, harry and hetty…and li’l puff
- in the first 3 minutes…
- Not in stellar fusion…
- in the first 3 minutes…
li’l puff because that’s what happens to lithium in star forges; it turns to ‘puff’…
So, free range lithium is a refugee from the big bang, a victim of genocide by stars…plus small amounts made by cosmic ray spallation — the same process that makes boron and beryllium…
This is why lithium is relatively rare despite being element 3…
- the universe made it first and has been destroying it ever since…
- The oldest element that’s been running from its own creator for 13.8 billion years…
- And now we mine it for batteries...to meet its creator…
The element that survived the Big Bang and billions of years of stellar destruction ends up in my iPad...
- so I can write its eulogy…
Appendix 4
Atomic number
Atomic number, not atomic weight
- runs from 1 to 92.
Atomic number = the number of protons in the nucleus.
- this defines the element…
- Hydrogen has 1 proton…
- Helium has 2…
- Lithium has 3…
- Carbon has 6…
- Iron has 26….
- Uranium has 92…
The atomic number IS the element’s identity…
So, the fusion ladder in climbing atomic numbers:
∙ Hydrogen: 1 proton
∙ Helium: 2 protons
∙ Carbon: 6 protons
∙ Neon: 10
∙ Oxygen: 8
∙ Magnesium: 12
∙ Silicon: 14
∙ Sulphur: 16
∙ Iron: 26
Notice the jumps aren’t sequential
- Hydrogen to helium is 1→2.
- helium to carbon is 2→6 (three heliums fusing).
The triple alpha process skips 3, 4, 5 entirely…
Not a product of stellar fusion…in fact.. destroyed by it…are…
∙ Lithium: 3 protons
∙ beryllium: 4 protons
∙ boron: 5 protons…
The 92 naturally occurring elements fill atomic numbers 1 to 92, but the universe didn’t make them in order.
It made them by whatever fusion or neutron capture process the physics allowed, skipping around the periodic table like a jazz musician, not a scales player.
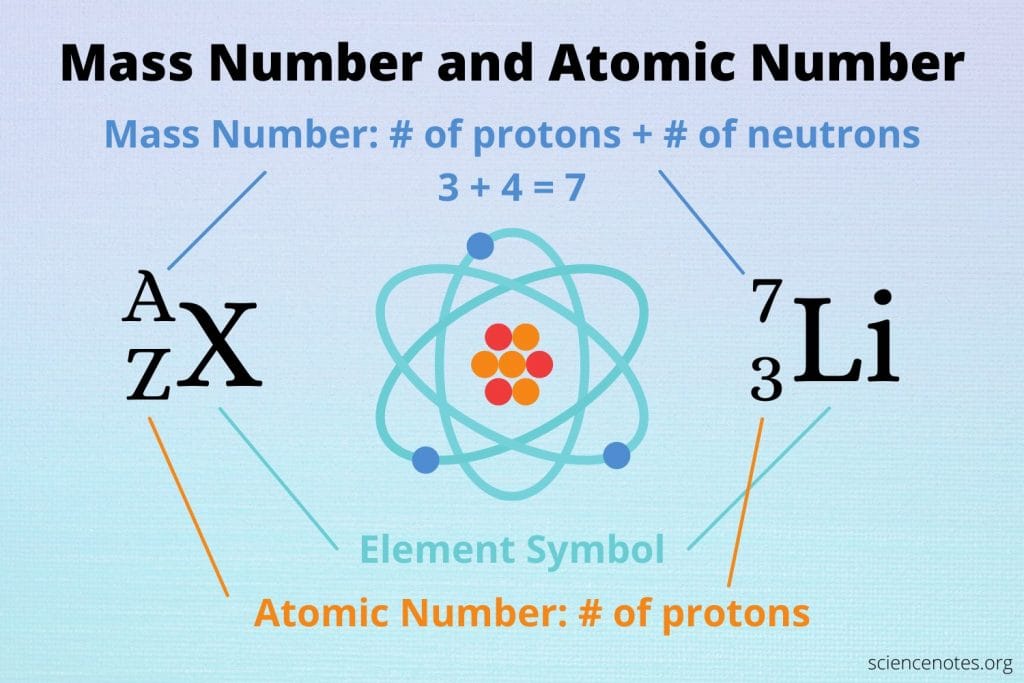
Atomic weight
Atomic weight (or atomic mass) is different…
- it’s the total number of protons plus neutrons.
- This varies because elements can have different numbers of neutrons (isotopes).
- Carbon-12 has 6 protons + 6 neutrons = atomic weight 12.
- Carbon-14 has 6 protons + 8 neutrons = atomic weight 14.
- Still carbon
- same atomic number, different weight.
- Lithium-7 (the most common isotope from the Big Bang) has 3 protons + 4 neutrons = atomic weight 7…see in graphic above…
- Lithium-6 has 3 protons + 3 neutrons = atomic weight 6. Rarer…
- Both are lithium because both have 3 protons.
The neutron count changes the weight but not the identity...
Appendix 5
Heat death of the universe
In about 10^100 years a googol,
- the Internet will still be running as Google;
- all hydrogen will be consumed…
- no new stars can form...
- Existing stars burn out...
- White dwarfs cool to black dwarfs...
- Neutron stars fade...
- Black holes evaporate via Hawking radiation...
The universe reaches maximum entropy
- uniform temperature,
- no energy gradients…
- no work possible...
- No structure...
- No chemistry...
- No life…
- cold, dark…
- expanding nothing...
- cold, dark…